Lithium-ion batteries can be safer and more compact if they contain a solid that conducts the ions, rather than a liquid. However, no suitable solids have been discovered that would allow the battery to maintain stable performance. Therefore, researchers are eagerly searching for potential new materials. A new material with promising ionic permeability properties has been discovered at the University of Liverpool. It's Friday In the scientific journal Sciences.
Lithium-ion batteries are used, for example, in electric bicycles, cars and phones. A chemical reaction always occurs in batteries: lithium ions move from the anode to the cathode when discharging and back again when charging. Their path passes through the electrolyte: a substance between the anode and cathode that allows ions to pass, but not electrons. The electrons are forced to move outside, so the battery provides energy.
Fire risk is a major disadvantage. The culprit is the flammable liquid electrolyte. It requires a strong metal casing around the battery and a separating layer – a separator – inside the battery.
Less space and weight
The solid is an ionic conductor and is non-flammable. This makes it safer, and in its wake other parts of the battery can also be improved. “The solid electrolyte takes up less space and weight than the liquid electrolyte, no longer requires a separator and requires less metal packing around the battery cell,” says Mark Hoegbein, professor of nanomaterials for energy conversion and storage at the University of Twente. “But the most important thing is that the current graphite anode could potentially be replaced with pure lithium metal. This would significantly reduce the weight and size of each battery cell.
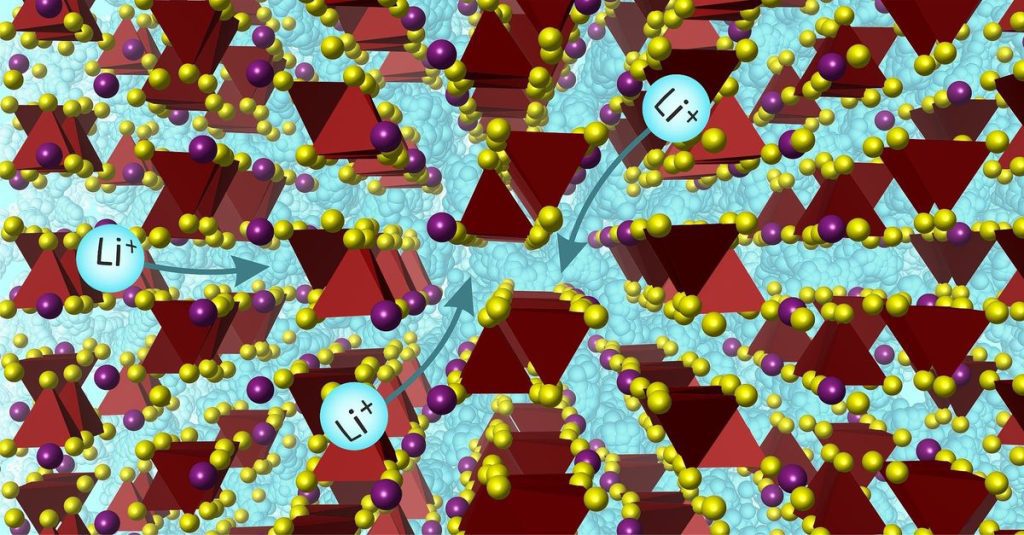
Finding a suitable material is difficult. “It is not at all easy for a solid to allow a charged atom, or ion, to move through its atomic structure if the space for movement is very limited,” Hoegbein summarizes the difficulty.
The new material proposed by Liverpool-Lee researchers7bad2s7I – has an ionic conductivity similar to a liquid electrolyte. In addition to the specific composition, the material also has a specific atomic structure, developed by combining experimental chemical research and artificial intelligence.
“It's an interesting result with a new material with promising properties,” says Hoegbein. “But the question is always: How stable is a battery cell? Over the past decade, several classes of materials have been developed with promising performance. With other sulfides with values similar to liquid electrolytes, significant degradation occurs. These batteries can only be charged and discharged for a limited number of “There are other substances that decompose less, but have less ionic conductivity.”
Researchers agree Sciences Lee's stability is not discussed in detail7bad2s71. In particular, they emphasize that their discovery could lead to a new family of electrolyte materials.

“Coffee buff. Twitter fanatic. Tv practitioner. Social media advocate. Pop culture ninja.”










More Stories
Which can cause an increase in nitrogen.
The Central State Real Estate Agency has no additional space to accommodate Ukrainians.
The oystercatcher, the “unlucky national bird,” is increasingly breeding on rooftops.